Kolekce 35+ Atom Economy Equation Zdarma
Kolekce 35+ Atom Economy Equation Zdarma. % atom economy = (6 / 34) * 100 = 17.7% ; In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Whenever there is only one product, the atom economy will always be 100%. It is found directly from the balanced equation by calculating the mr of the desired product.
Nejchladnější Solved One Measure Of Green Chemistry Is Atom Economy For Chegg Com
Whenever there is only one product, the atom economy will always be 100%. Calculate the relative molecular mass of each of the products. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. Electrolysis of water is when water is converted into …Calculate the relative molecular mass of each of the products.
Whenever there is only one product, the atom economy will always be 100%. Atom economy = 100 x. Mass of desired useful product. Construct a chemical equation for the given reaction. 08.01.2018 · then, calculate the % atom economy: Calculate the relative molecular mass of each of the products. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.. Electrolysis of water is when water is converted into … The rest of the atoms or mass is wasted. Green chemists define atom economy as: The percentage atom economy of a reaction is calculated using this equation: How to calculate atom economy step 1. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 08.01.2018 · then, calculate the % atom economy:.. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6.

Calculate the percentage atom economy. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. It is found directly from the balanced equation by calculating the mr of the desired product. The atom economy can be calculated in either of two ways: How to calculate atom economy step 1. Atom economy = 100 x. % atom economy = (6 / 34) * 100 = 17.7% ; % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% Electrolysis of water is when water is converted into … The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product.. Reactants desired product + waste products. It is found directly from the balanced equation by calculating the mr of the desired product. Its sometimes referred to as atom utilisation. Electrolysis of water is when water is converted into … The rest of the atoms or mass is wasted. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Mass of desired useful product. Write out the balanced equation. Calculate the relative molecular mass of each of the products.

Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6.. Calculate the relative molecular mass of each of the products. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Its sometimes referred to as atom utilisation. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The atom economy can be calculated in either of two ways: For the general chemical reaction:
Electrolysis of water is when water is converted into …. Mass of desired useful product. How to calculate atom economy step 1. Write out the balanced equation. The atom economy can be calculated in either of two ways: In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. For the general chemical reaction: The rest of the atoms or mass is wasted.
% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Green chemists define atom economy as: Calculate the percentage atom economy. Mass of desired useful product. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. It is found directly from the balanced equation by calculating the mr of the desired product. % atom economy = (6 / 34) * 100 = 17.7% ; It is found directly from the balanced equation by calculating the mr of the desired product... Mass of desired useful product.

Its sometimes referred to as atom utilisation. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Whenever there is only one product, the atom economy will always be 100%.. Write out the balanced equation.

Its sometimes referred to as atom utilisation... The atom economy can be calculated in either of two ways:. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.

Calculate the relative molecular mass of each of the products... Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Electrolysis of water is when water is converted into … It is found directly from the balanced equation by calculating the mr of the desired product. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Mass of desired useful product... It is found directly from the balanced equation by calculating the mr of the desired product.

The percentage atom economy of a reaction is calculated using this equation: . In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product.

Reactants desired product + waste products. 08.01.2018 · then, calculate the % atom economy: Whenever there is only one product, the atom economy will always be 100%. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: The atom economy can be calculated in either of two ways: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The percentage atom economy of a reaction is calculated using this equation: How to calculate atom economy step 1. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. It is found directly from the balanced equation by calculating the mr of the desired product.

For the general chemical reaction: .. Write out the balanced equation.

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Write out the balanced equation. It is found directly from the balanced equation by calculating the mr of the desired product. How to calculate atom economy step 1. It is found directly from the balanced equation by calculating the mr of the desired product. The atom economy can be calculated in either of two ways: Electrolysis of water is when water is converted into … Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. % atom economy = (6 / 34) * 100 = 17.7% ; Green chemists define atom economy as:

Calculate the relative molecular mass of each of the products.. Mass of desired useful product. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. 08.01.2018 · then, calculate the % atom economy: It is found directly from the balanced equation by calculating the mr of the desired product. Its sometimes referred to as atom utilisation. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product... Electrolysis of water is when water is converted into …

Reactants desired product + waste products. Calculate the relative molecular mass of each of the products. For the general chemical reaction: The rest of the atoms or mass is wasted. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. It is found directly from the balanced equation by calculating the mr of the desired product. Mass of desired useful product. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. Atom economy = 100 x. The rest of the atoms or mass is wasted.
The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. 08.01.2018 · then, calculate the % atom economy: Calculate the relative molecular mass of each of the products. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Electrolysis of water is when water is converted into … 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: How to calculate atom economy step 1. Its sometimes referred to as atom utilisation. Construct a chemical equation for the given reaction.. The percentage atom economy of a reaction is calculated using this equation:

The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (6 / 34) * 100 = 17.7% ;

Whenever there is only one product, the atom economy will always be 100%. .. The rest of the atoms or mass is wasted.

In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. For the general chemical reaction: How to calculate atom economy step 1. Reactants desired product + waste products. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. 08.01.2018 · then, calculate the % atom economy: The atom economy can be calculated in either of two ways: Calculate the percentage atom economy. How to calculate atom economy step 1.

Construct a chemical equation for the given reaction. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. Calculate the relative molecular mass of each of the products. Calculate the percentage atom economy. It is found directly from the balanced equation by calculating the mr of the desired product.

The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Reactants desired product + waste products. The percentage atom economy of a reaction is calculated using this equation: The atom economy can be calculated in either of two ways: Green chemists define atom economy as: Mass of desired useful product.

Mass of desired useful product.. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Calculate the relative molecular mass of each of the products. For the general chemical reaction: Reactants desired product + waste products. % atom economy = (6 / 34) * 100 = 17.7% ; Atom economy = 100 x. Construct a chemical equation for the given reaction. Whenever there is only one product, the atom economy will always be 100%. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. Its sometimes referred to as atom utilisation.. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products.
For the general chemical reaction:.. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Mass of desired useful product. Write out the balanced equation. Electrolysis of water is when water is converted into ….. Atom economy = 100 x.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% Its sometimes referred to as atom utilisation. Mass of desired useful product.. Its sometimes referred to as atom utilisation.

The percentage atom economy of a reaction is calculated using this equation: Construct a chemical equation for the given reaction. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Calculate the percentage atom economy. It is found directly from the balanced equation by calculating the mr of the desired product.
The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. Construct a chemical equation for the given reaction. The rest of the atoms or mass is wasted. Atom economy = 100 x. Calculate the relative molecular mass of each of the products. Mass of desired useful product. How to calculate atom economy step 1... Write out the balanced equation.

Whenever there is only one product, the atom economy will always be 100%. Reactants desired product + waste products. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. It is found directly from the balanced equation by calculating the mr of the desired product. It is found directly from the balanced equation by calculating the mr of the desired product. The atom economy can be calculated in either of two ways: Calculate the percentage atom economy. Calculate the percentage atom economy.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. Its sometimes referred to as atom utilisation.. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.

Its sometimes referred to as atom utilisation... Calculate the percentage atom economy. The percentage atom economy of a reaction is calculated using this equation: Its sometimes referred to as atom utilisation. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: Mass of desired useful product. 08.01.2018 · then, calculate the % atom economy: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% For the general chemical reaction:

Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. Write out the balanced equation.. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6.

It is found directly from the balanced equation by calculating the mr of the desired product. 08.01.2018 · then, calculate the % atom economy: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Its sometimes referred to as atom utilisation. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: Electrolysis of water is when water is converted into … Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Write out the balanced equation. Green chemists define atom economy as: For the general chemical reaction: Whenever there is only one product, the atom economy will always be 100%... Whenever there is only one product, the atom economy will always be 100%.

Its sometimes referred to as atom utilisation. Calculate the percentage atom economy. Electrolysis of water is when water is converted into … Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. It is found directly from the balanced equation by calculating the mr of the desired product. % atom economy = (6 / 34) * 100 = 17.7% ; Mass of desired useful product. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6... The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

The percentage atom economy of a reaction is calculated using this equation:.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · then, calculate the % atom economy: Green chemists define atom economy as: % atom economy = (6 / 34) * 100 = 17.7% ; Reactants desired product + waste products. For the general chemical reaction: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Whenever there is only one product, the atom economy will always be 100%... Its sometimes referred to as atom utilisation.

In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the.. % atom economy = (6 / 34) * 100 = 17.7% ; Whenever there is only one product, the atom economy will always be 100%. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.. The rest of the atoms or mass is wasted.

The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. 08.01.2018 · then, calculate the % atom economy: In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Its sometimes referred to as atom utilisation. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. 08.01.2018 · then, calculate the % atom economy:

The percentage atom economy of a reaction is calculated using this equation: Its sometimes referred to as atom utilisation. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. The rest of the atoms or mass is wasted. Green chemists define atom economy as: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: 08.01.2018 · then, calculate the % atom economy: Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6.

It is found directly from the balanced equation by calculating the mr of the desired product. For the general chemical reaction: Whenever there is only one product, the atom economy will always be 100%. How to calculate atom economy step 1. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. The rest of the atoms or mass is wasted. Mass of desired useful product.. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6.

Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Its sometimes referred to as atom utilisation. How to calculate atom economy step 1. Green chemists define atom economy as: Write out the balanced equation.. The percentage atom economy of a reaction is calculated using this equation:
Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. How to calculate atom economy step 1. Electrolysis of water is when water is converted into … For the general chemical reaction: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The percentage atom economy of a reaction is calculated using this equation: Atom economy = 100 x. 08.01.2018 · then, calculate the % atom economy: It is found directly from the balanced equation by calculating the mr of the desired product. Write out the balanced equation.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100%

For the general chemical reaction:. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. Reactants desired product + waste products. Green chemists define atom economy as: Its sometimes referred to as atom utilisation. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Mass of desired useful product. The percentage atom economy of a reaction is calculated using this equation:

In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the.. It is found directly from the balanced equation by calculating the mr of the desired product.

Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. 08.01.2018 · then, calculate the % atom economy: Calculate the percentage atom economy. Construct a chemical equation for the given reaction.
In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. Mass of desired useful product. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. 08.01.2018 · then, calculate the % atom economy: Calculate the relative molecular mass of each of the products. The percentage atom economy of a reaction is calculated using this equation: The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Green chemists define atom economy as:. The atom economy can be calculated in either of two ways:

Electrolysis of water is when water is converted into ….. Write out the balanced equation.. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.

In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. For the general chemical reaction: Atom economy = 100 x. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: How to calculate atom economy step 1. The atom economy can be calculated in either of two ways: Its sometimes referred to as atom utilisation. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6.

Reactants desired product + waste products. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: Calculate the relative molecular mass of each of the products. Green chemists define atom economy as: In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. Whenever there is only one product, the atom economy will always be 100%. 08.01.2018 · then, calculate the % atom economy: The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. The atom economy can be calculated in either of two ways: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

Calculate the percentage atom economy. % atom economy = (6 / 34) * 100 = 17.7% ; Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.

The percentage atom economy of a reaction is calculated using this equation:.. .. Mass of desired useful product.

Its sometimes referred to as atom utilisation. Calculate the percentage atom economy. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. For the general chemical reaction: It is found directly from the balanced equation by calculating the mr of the desired product. Whenever there is only one product, the atom economy will always be 100%. The rest of the atoms or mass is wasted. Mass of desired useful product.. The atom economy can be calculated in either of two ways:

Calculate the relative molecular mass of each of the products. .. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6.

01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: For the general chemical reaction: How to calculate atom economy step 1. Atom economy = 100 x. It is found directly from the balanced equation by calculating the mr of the desired product. % atom economy = (6 / 34) * 100 = 17.7% ; In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: Construct a chemical equation for the given reaction... Green chemists define atom economy as:

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% % atom economy = (6 / 34) * 100 = 17.7% ; The rest of the atoms or mass is wasted. It is found directly from the balanced equation by calculating the mr of the desired product.. Write out the balanced equation.

Calculate the relative molecular mass of each of the products. Electrolysis of water is when water is converted into … Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table... It is found directly from the balanced equation by calculating the mr of the desired product.

Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Mass of desired useful product. Write out the balanced equation. Calculate the percentage atom economy. The percentage atom economy of a reaction is calculated using this equation: Construct a chemical equation for the given reaction. Calculate the relative molecular mass of each of the products. Atom economy = 100 x. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6.

08.01.2018 · then, calculate the % atom economy:.. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: Reactants desired product + waste products. Construct a chemical equation for the given reaction. How to calculate atom economy step 1. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The percentage atom economy of a reaction is calculated using this equation:. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

Its sometimes referred to as atom utilisation. Construct a chemical equation for the given reaction. Atom economy = 100 x. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.. Electrolysis of water is when water is converted into …

It is found directly from the balanced equation by calculating the mr of the desired product. Atom economy = \(\frac{\textup{total m}_{r} \textup{of the desired product}}{\textup{total m}_{r} \textup{of all reactants}}\) × 100 atom economy = \(\frac{6}{34} \times 100\) atom economy = 17.6. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the.

It is found directly from the balanced equation by calculating the mr of the desired product. . Atom economy = 100 x.

Calculate the percentage atom economy... Reactants desired product + waste products. It is found directly from the balanced equation by calculating the mr of the desired product. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Write out the balanced equation. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% Atom economy = 100 x. Calculate the relative molecular mass of each of the products. Whenever there is only one product, the atom economy will always be 100%.. % atom economy = (6 / 34) * 100 = 17.7% ;

Atom economy = 100 x... Calculate the percentage atom economy. The atom economy can be calculated in either of two ways: Its sometimes referred to as atom utilisation. Whenever there is only one product, the atom economy will always be 100%. Reactants desired product + waste products. The percentage atom economy of a reaction is calculated using this equation: For the general chemical reaction: % atom economy = (6 / 34) * 100 = 17.7% ; Mass of desired useful product.
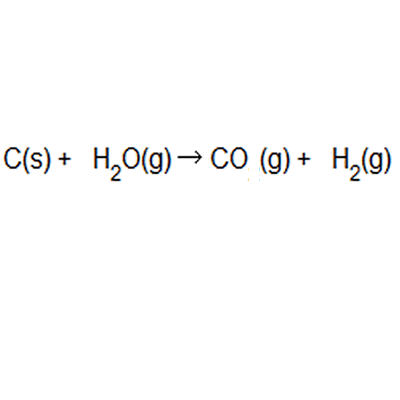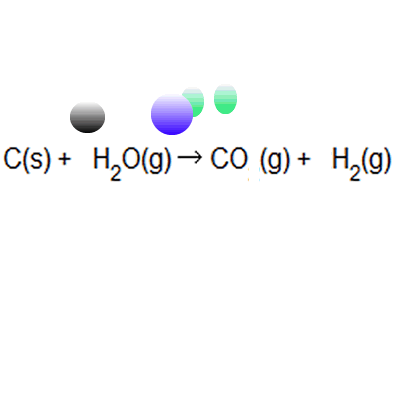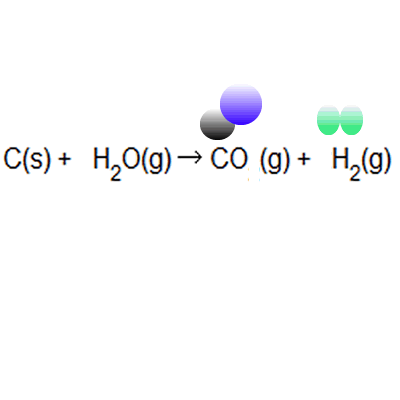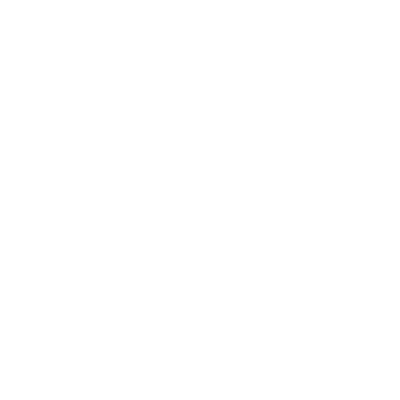
% atom economy = (6 / 34) * 100 = 17.7% ; Calculate the percentage atom economy. Write out the balanced equation. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Mass of desired useful product. 08.01.2018 · then, calculate the % atom economy: Reactants desired product + waste products. Its sometimes referred to as atom utilisation. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the desired product. % atom economy = (6 / 34) * 100 = 17.7% ;. Its sometimes referred to as atom utilisation.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/137) x 100 = 100% .. Electrolysis of water is when water is converted into …

Green chemists define atom economy as: Construct a chemical equation for the given reaction. Reactants desired product + waste products. It is found directly from the balanced equation by calculating the mr of the desired product. Write out the balanced equation. 01.07.2020 · a general way to proceed in order to calculate the atom economy is to use the following steps: The rest of the atoms or mass is wasted. The atom economyof a reaction is a theoretical percentage measure of the amount of starting materials that ends up as the 'desired' useful reaction products. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product... The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.
